In recent years, the pursuit of efficient and sustainable energy sources has driven extensive research into solar photovoltaic materials, which are pivotal for converting sunlight into electricity through the photovoltaic effect. As a researcher in this field, I have observed remarkable progress across various material systems, each contributing to the evolution of solar systems worldwide. This article aims to provide a comprehensive overview of the developments in both inorganic and organic photovoltaic materials, with a focus on their properties, performance, and integration into modern solar systems. I will incorporate tables and mathematical formulations to summarize key findings, while emphasizing the importance of material innovation for advancing solar system technologies. The keyword ‘solar system’ will be frequently referenced to highlight the interconnectedness of material science and photovoltaic applications, ensuring that the discussion remains grounded in real-world energy solutions.
The foundation of solar photovoltaic technology lies in materials that can efficiently absorb sunlight and generate charge carriers. Ideally, semiconductor materials for solar cells should have a bandgap between 1.1 eV and 1.7 eV to maximize photon absorption across the solar spectrum, exhibit high photoconversion efficiency, be environmentally benign, and allow for scalable production with long-term stability. Over the decades, solar cell materials have evolved through three generations, each marked by improvements in efficiency, cost, and manufacturability. The first generation relies on crystalline silicon, which has dominated the market due to its mature technology and relatively high efficiencies. However, challenges such as material scarcity and high costs have spurred the development of second-generation thin-film materials and third-generation concepts that leverage novel structures and composites. In this review, I will delve into the specifics of these materials, using analytical tools like efficiency equations and comparative tables to elucidate their roles in enhancing solar system performance.
To quantify the performance of photovoltaic materials, the photoconversion efficiency ($\eta$) is a critical metric, defined as:
$$ \eta = \frac{P_{\text{out}}}{P_{\text{in}}} \times 100\% $$
where $P_{\text{out}}$ is the electrical power output from the solar cell and $P_{\text{in}}$ is the incident solar power. This formula underpins much of the research discussed here, as scientists strive to maximize $\eta$ through material optimization. Additionally, the bandgap energy ($E_g$) influences the theoretical maximum efficiency, often described by the Shockley-Queisser limit, which for a single-junction solar cell under standard conditions is approximately 33%. Innovations in material design aim to approach or exceed this limit, thereby improving the overall efficacy of solar systems.
Inorganic photovoltaic materials have been the cornerstone of early solar systems, offering robustness and proven efficiency. Silicon-based materials, including monocrystalline, polycrystalline, and amorphous silicon, are widely used. Monocrystalline silicon cells, for instance, have achieved efficiencies up to 26.7% in laboratory settings, thanks to their high purity and ordered crystal structure. The photocurrent density ($J_{sc}$) in such cells can be enhanced through nanostructuring, as demonstrated in hybrid designs combining silicon nanowires with organic polymers. For example, integrating single-crystal silicon nanowires with poly(3-hexylthiophene)-[6,6]-phenyl-C61-butyric acid methyl ester has boosted $J_{sc}$ from 7.17 mA/cm² to 11.61 mA/cm², illustrating how material engineering can optimize light absorption and charge collection in solar systems. Polycrystalline silicon, while more cost-effective, suffers from grain boundaries that reduce carrier lifetimes, leading to efficiencies around 17–20%. Amorphous silicon alloys have emerged as flexible alternatives, with efficiencies improving through hydrogenation and doping strategies.
Beyond silicon, compound semiconductors like gallium arsenide (GaAs), cadmium telluride (CdTe), and copper zinc tin sulfide (Cu₂ZnSnS₄) have gained attention for their superior optical properties and potential for high-efficiency solar systems. GaAs, with a direct bandgap of 1.42 eV, is particularly suited for concentrated photovoltaic applications, achieving efficiencies exceeding 29% in multi-junction configurations. The open-circuit voltage ($V_{oc}$) in GaAs-based cells can reach up to 1.1 V, as seen in nanostructured designs that minimize recombination losses. However, concerns over toxicity and resource availability have driven research into abundant and non-toxic alternatives. For instance, Cu₂ZnSnS₄ (CZTS) nanocrystals have shown promise with efficiencies around 0.74% under AM1.5G illumination, though further optimization is needed to compete with established materials. To summarize the properties of key inorganic materials, I present Table 1, which compares their bandgaps, typical efficiencies, and relevance to solar system integration.
| Material | Bandgap (eV) | Typical Efficiency (%) | Advantages for Solar Systems |
|---|---|---|---|
| Monocrystalline Si | 1.12 | 20–26.7 | High stability, mature technology |
| Polycrystalline Si | 1.1–1.2 | 17–20 | Lower cost, scalable production |
| Amorphous Si | 1.7–1.8 | 10–12 | Flexibility, low-temperature processing |
| GaAs | 1.42 | 28–29 | High efficiency, radiation resistance |
| CdTe | 1.45 | 22–23 | Low-cost thin films, good absorption |
| CZTS | 1.4–1.5 | 0.7–12.6 | Abundant elements, non-toxic potential |
Metal oxides like titanium dioxide (TiO₂) and zinc oxide (ZnO) have also played significant roles, especially in dye-sensitized solar cells (DSSCs), which offer low-cost alternatives for niche solar system applications. TiO₂, with a bandgap of 3.2 eV, requires sensitizers to absorb visible light, but its high surface area and electron transport properties make it ideal for mesoporous electrodes. The efficiency of TiO₂-based DSSCs can be modeled using the incident photon-to-current efficiency (IPCE), given by:
$$ \text{IPCE}(\lambda) = \frac{1240 \times J_{sc}(\lambda)}{\lambda \times P_{\text{in}}(\lambda)} $$
where $\lambda$ is the wavelength in nanometers. ZnO, with a similar bandgap but higher electron mobility (up to 200 cm²/V·s), has been used in nanowire arrays that enhance light scattering and dye loading, leading to efficiencies around 5–7% in optimized devices. Another emerging material, tin sulfide (SnS), has an indirect bandgap of approximately 1.3 eV and high absorption coefficients (>10⁴ cm⁻¹), making it suitable for thin-film solar systems. Electrodeposited p-type SnS films have shown photoelectrochemical activity with an optical bandgap of 1.17 eV, highlighting its potential for sustainable photovoltaic applications.
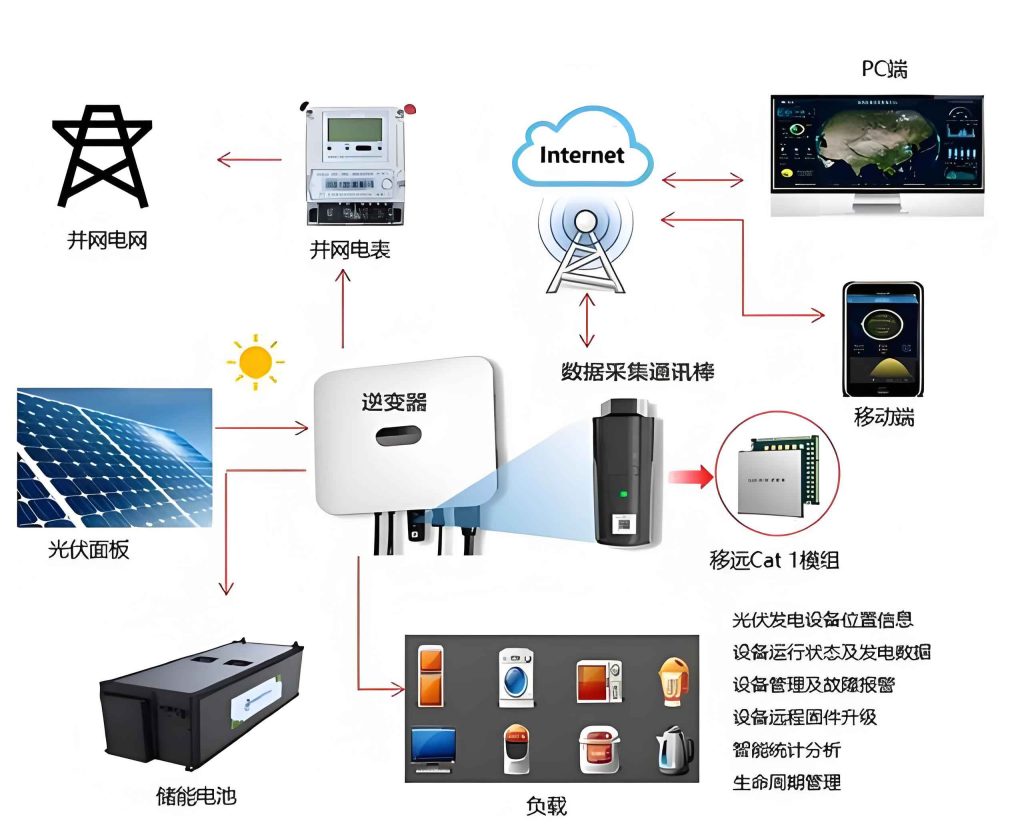
Organic photovoltaic materials, on the other hand, offer advantages such as tunable electronic properties, lightweight design, and solution processability, which can facilitate the deployment of flexible and portable solar systems. Phthalocyanine dyes, for example, are p-type semiconductors with strong absorption in the near-infrared region (around 700 nm), aligning well with the solar spectrum. Metal phthalocyanines (MPcs), where M can be copper or zinc, have been used as donors in bilayer solar cells, achieving efficiencies up to 5% when combined with fullerene acceptors. The charge separation dynamics in these systems can be described by the electron transfer rate constant ($k_{et}$), derived from Marcus theory:
$$ k_{et} = \frac{2\pi}{\hbar} |V|^2 \frac{1}{\sqrt{4\pi\lambda k_B T}} \exp\left(-\frac{(\Delta G + \lambda)^2}{4\lambda k_B T}\right) $$
where $V$ is the electronic coupling, $\lambda$ is the reorganization energy, $\Delta G$ is the Gibbs free energy change, $k_B$ is Boltzmann’s constant, and $T$ is temperature. This equation helps in designing organic materials for efficient charge generation in solar systems.
Fullerene derivatives, particularly [6,6]-phenyl-C61-butyric acid methyl ester (PCBM), are common acceptors in organic solar cells due to their high electron affinity and ability to form percolation networks for charge transport. The power conversion efficiency of PCBM-based devices has surpassed 10% in recent years, with open-circuit voltages around 0.8–0.9 V. However, their weak absorption in the visible range necessitates the use of complementary donors. Graphene-based materials have emerged as a frontier in this domain, offering exceptional conductivity, mechanical strength, and transparency. Pristine graphene has a zero bandgap, but functionalization with moieties like porphyrin can induce a tunable bandgap and enhance optical properties. For instance, covalent functionalization of graphene with porphyrin has led to hybrid materials with improved nonlinear optical characteristics, beneficial for advanced solar system components. Chemical doping of graphene with bis(trifluoromethanesulfonyl)amide has increased solar cell efficiencies from 4.5% to 8.6% under AM1.5 conditions, demonstrating the potential of graphene in next-generation photovoltaic devices.
To compare organic materials, Table 2 outlines their key parameters and contributions to solar system performance. This includes metrics like highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels, which influence the open-circuit voltage through the relation $V_{oc} \approx \frac{1}{e}(|\text{HOMO}_{\text{donor}}| – |\text{LUMO}_{\text{acceptor}}|) – 0.3$ V, where $e$ is the elementary charge.
| Material | Type | HOMO (eV) | LUMO (eV) | Efficiency (%) | Role in Solar Systems |
|---|---|---|---|---|---|
| Zinc Phthalocyanine | Donor | -5.2 | -3.5 | 4–5 | Strong absorption, p-type semiconductor |
| PCBM | Acceptor | -6.1 | -3.7 | 10–11 | High electron mobility, good blendability |
| Porphyrin-Graphene | Hybrid | -4.8 | -3.9 | 8–9 | Enhanced charge transfer, tunable bandgap |
| Poly(3-hexylthiophene) | Donor | -5.0 | -3.0 | 6–7 | Solution processable, flexible films |
The development of three-dimensional graphene structures, such as honeycomb-like frameworks, has further expanded the possibilities for solar system integration. These 3D graphenes exhibit high surface area, excellent electrical conductivity, and catalytic activity, potentially replacing expensive platinum in counter-electrodes for DSSCs. The efficiency of such systems can be approximated using the fill factor (FF), defined as:
$$ \text{FF} = \frac{V_{mp} \times J_{mp}}{V_{oc} \times J_{sc}} $$
where $V_{mp}$ and $J_{mp}$ are the voltage and current density at maximum power point. For 3D graphene-based electrodes, FF values exceeding 0.75 have been reported, contributing to overall efficiencies above 10%. This underscores how material innovation can directly enhance the performance metrics of solar systems, from residential panels to large-scale photovoltaic farms.
In addition to individual materials, hybrid and composite systems are gaining traction for their ability to synergize the strengths of inorganic and organic components. For example, perovskite solar cells, which incorporate organic-inorganic lead halide materials, have achieved efficiencies over 25%, rivaling silicon-based technologies. The photocurrent in these devices can be modeled using the diode equation modified for photovoltaic effects:
$$ J = J_{ph} – J_0 \left[\exp\left(\frac{e(V + J R_s)}{n k_B T}\right) – 1\right] – \frac{V + J R_s}{R_{sh}} $$
where $J_{ph}$ is the photogenerated current density, $J_0$ is the reverse saturation current, $R_s$ is series resistance, $R_{sh}$ is shunt resistance, and $n$ is the ideality factor. Such formulations are essential for optimizing device architectures in complex solar systems. Moreover, tandem solar cells, which stack multiple absorbers with complementary bandgaps, can surpass the Shockley-Queisser limit by better utilizing the solar spectrum. The theoretical efficiency of a tandem cell with $N$ junctions is given by:
$$ \eta_{\text{tandem}} = \sum_{i=1}^{N} \eta_i \left(\frac{E_{g,i}}{E_{\text{sun}}}\right) $$
where $\eta_i$ is the efficiency of the $i$-th junction, $E_{g,i}$ is its bandgap, and $E_{\text{sun}}$ is the total solar irradiance. This approach is pivotal for advancing high-efficiency solar systems in space and terrestrial applications.
Looking ahead, the future of solar photovoltaic materials will likely focus on several key directions: enhancing conversion efficiencies beyond 30% through multi-junction and spectral splitting designs; reducing manufacturing costs via scalable methods like roll-to-roll printing; improving material sustainability by using abundant, non-toxic elements; and integrating smart functionalities such as self-healing or adaptive optics. For instance, quantum dot solar cells leverage size-dependent bandgaps to tailor absorption profiles, with efficiencies projected to reach 20% in the coming years. The quantum confinement effect can be described by the Brus equation for spherical nanoparticles:
$$ E_g^{\text{nano}} = E_g^{\text{bulk}} + \frac{\hbar^2 \pi^2}{2R^2} \left(\frac{1}{m_e^*} + \frac{1}{m_h^*}\right) – \frac{1.8 e^2}{4\pi\varepsilon R} $$
where $R$ is the nanoparticle radius, $m_e^*$ and $m_h^*$ are effective masses of electrons and holes, and $\varepsilon$ is the dielectric constant. This tunability makes quantum dots promising for customized solar systems in diverse environments.
In conclusion, the progress in solar photovoltaic materials has been instrumental in driving the global adoption of solar systems as a clean energy source. From inorganic workhorses like silicon and GaAs to organic innovators like phthalocyanines and graphene, each material class offers unique advantages that can be harnessed through careful design and integration. As research continues to push the boundaries of efficiency, durability, and cost-effectiveness, I am confident that these advancements will pave the way for more resilient and widespread solar systems, contributing to a sustainable energy future. The interplay between material science and system engineering remains crucial, and ongoing efforts in hybrid materials, nanostructuring, and computational modeling will undoubtedly yield further breakthroughs, ensuring that solar systems play a central role in meeting the world’s energy demands.
