The rapid development of clean energy has placed significant demands on grid stability, making battery energy storage systems a critical component for peak shaving and renewable energy integration. The safety of lithium-ion batteries, however, remains a paramount concern that hinders the wider adoption of large-scale battery energy storage system deployments. Thermal runaway incidents, often originating from a single cell, can cascade through an entire system, leading to catastrophic failure. This study focuses on investigating the early warning signs of thermal failure in a commercial 100 Ah Lithium Iron Phosphate (LFP) cell, a common chemistry in stationary battery energy storage system applications, subjected to overcharge abuse.
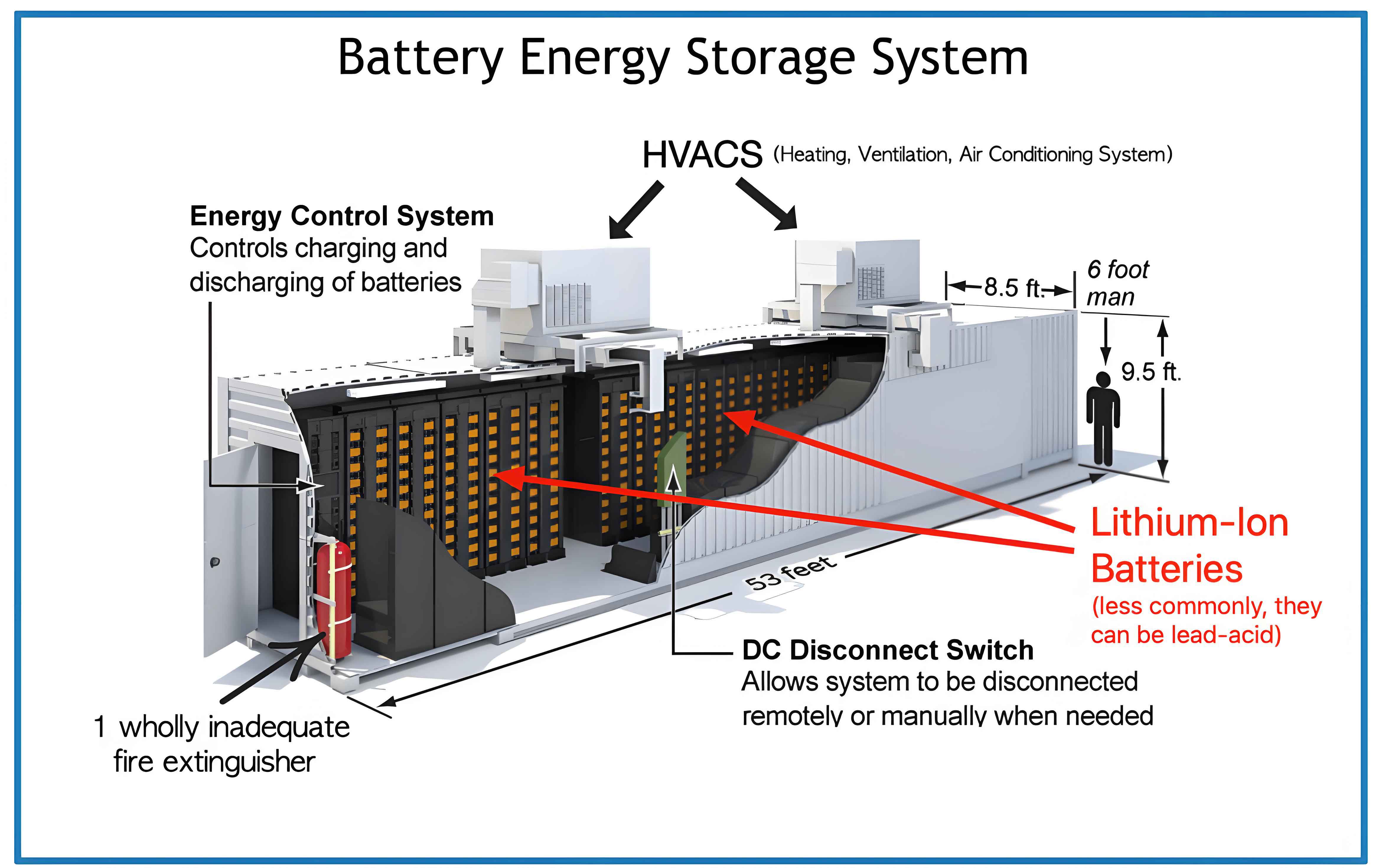
To explore the characteristic parameters leading to thermal failure, we employed a two-stage overcharge protocol. This method allows us to probe the battery’s behavior in the pre-failure stage and assess its safety state after charging intervention. The experimental cell specifications are detailed below.
| Parameter | Value |
|---|---|
| Rated Capacity (Ah) | 100 |
| Nominal Voltage Range (V) | 2.5 – 3.65 |
| Internal Impedance (1 kHz) (mΩ) | 0.2 – 0.5 |
| Mass (kg) | 2.0 |
| Electrolyte | LiPF6 in EC/EMC/DEC/DMC |
The core of our experimental setup involved a semi-enclosed chamber housing the cell, coupled with a comprehensive monitoring system. This system recorded voltage, current, and surface temperatures at multiple points. Crucially, we integrated real-time electrochemical gas detectors (for H2, CO, CO2) and used gas chromatography (GC) for detailed offline analysis of gas composition. This multi-parameter approach is essential for developing robust safety protocols for a battery energy storage system.
The experimental procedure was designed to simulate a late-stage failure scenario. In the first stage, the cell was charged at a 0.5C rate (50 A) until its voltage rapidly climbed to 20 V, a point we identified in pre-tests as immediately preceding thermal runaway. Charging was then halted for 30 minutes to observe the cell’s behavior post-intervention. In the second stage, charging was resumed at a lower 0.1C rate (10 A) to deliberately push the cell into thermal failure, allowing us to capture the final failure characteristics.
Experimental Observations and Parameter Analysis
The overcharge process revealed distinct stages in voltage evolution, as shown in our data. The initial stage involved a rapid voltage rise above the nominal cutoff, followed by a plateau of slow increase. A critical turning point was observed when the voltage began to drop, coinciding with visible cell swelling. Subsequently, the safety valve opened, releasing gas and electrolyte. Following this, the voltage entered a second, steeper rise phase. In our first-stage test, we interrupted charging at 20 V during this phase. The voltage profile for the second, failure-inducing stage is described by the following simplified relationship, where the rapid increase is driven by internal impedance rise:
$$ V(t) = V_0 + k_1 \cdot t \quad \text{(for t < t_{melt})} $$
$$ \text{At } t = t_{melt}, \quad V \rightarrow 0 \quad \text{(Internal Short Circuit)} $$
Where \( V_0 \) is the voltage at the start of the second-stage charge, \( k_1 \) is a high rate of increase, and \( t_{melt} \) is the time of separator meltdown leading to short circuit. Stopping the charge at 20 V in the first stage successfully prevented thermal runaway, demonstrating this as a viable late-stage voltage warning threshold. However, resuming charge after 30 minutes quickly led to failure with only a 1.5% SOC increment, indicating that the cell remained in a critical state.
Temperature measurements provided critical insights into internal reaction kinetics. After charging stopped in the first stage, surface temperatures continued to rise for approximately 5 minutes before gradually declining. This post-charge temperature rise is a key risk indicator, signifying that exothermic side reactions were continuing unabated using the accumulated lithium dendrites. The temperature gradient between the cell surface and its interior, inferred from gas evolution data, was significant. For instance, when C2H2 generation was detected (indicative of reactions starting around 200°C), the surface temperature was only 80-90°C. This large gradient, exceeding 110°C, highlights the limitation of surface-mounted sensors and underscores the need for internal parameter inference, such as through gas analysis, in a battery energy storage system safety design.
Gas generation was the most sensitive and early indicator of cell degradation. Our GC analysis provided a complete picture of the gas species evolved, as summarized in the table below. The data clearly shows the evolution from the first venting event to full thermal runaway.
| Sampling Point | Non-Hydrocarbon Gases (vol%) | Hydrocarbon Gases (vol%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO | CO2 | CH4 | C2H6 | C2H4 | C2H2 | C3H8 | C3H6 | C4H10 | |
| Pre-Venting | 0 | 0 | 0.1112 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Post-Vent (5 min) | 0.3596 | 0.0522 | 0.4284 | 0.0140 | 0.0078 | 0.0252 | 0.0004 | 0.0004 | 0.0002 | 0.0003 |
| Post-Vent (20 min) | 0.4724 | 0.0937 | 0.7002 | 0.0220 | 0.0134 | 0.1492 | 0.0009 | 0.0008 | 0.0012 | 0.0020 |
| Thermal Runaway | 7.4424 | 1.3725 | 3.8260 | 0.5523 | 0.1182 | 0.6224 | 0.0041 | 0.0259 | 0.0025 | 0.0180 |
The real-time detector data showed that H2 and CO were the first gases detected immediately after venting, with H2 rising fastest. GC analysis confirmed H2 as the most abundant gas throughout, constituting over 62% of the major gas species at its peak. Among hydrocarbons, C2H4 (ethylene) was dominant, making up 78.5% of hydrocarbon content at its peak before thermal runaway. The sequence and abundance make H2 the prime candidate for early warning.
Mechanistic Interpretation and Failure Pathways
The observed parameters can be explained by a cascade of internal electrochemical and chemical reactions. The initial overcharge leads to lithium plating and dendrite formation on the graphite anode. These dendrites react with the polyvinylidene fluoride (PVDF) binder, producing hydrogen gas and heat at relatively low temperatures:
$$ \text{2} \left( \text{–CH}_2\text{–CF}_2\text{–} \right) + \text{2Li} \rightarrow \left( \text{–CH=CF–} \right) + \text{LiF} + \text{H}_2 \quad \text{(1)} $$
Subsequently, lithium dendrites react with organic carbonate solvents (EC, DMC, EMC, DEC) in the electrolyte, generating various hydrocarbon gases (like C2H4, C2H6) and lithium alkyl carbonates. As temperature rises above approximately 90°C due to these exothermic reactions, the solid electrolyte interphase (SEI) decomposes. A key decomposition of lithium ethylene dicarbonate produces CO2, C2H4, and O2:
$$ (\text{CH}_2\text{OCO}_2\text{Li})_2 \rightarrow \text{Li}_2\text{CO}_3 + \text{C}_2\text{H}_4 + \frac{1}{2}\text{O}_2 + \text{CO}_2 \quad \text{(2)} $$
The released oxygen then reacts with the bulk electrolyte solvents, producing more CO2 (or CO under oxygen-starved conditions) and water, further accelerating heat generation. Concurrently, the delithiated LixFePO4 cathode becomes thermally unstable. The heat buildup eventually causes the polyolefin separator to melt and close its pores, drastically increasing internal resistance and triggering the final, steep voltage rise. Ultimately, separator collapse causes a large-scale internal short circuit, dumping the remaining energy and initiating thermal runaway.
The post-charge temperature rise is a critical finding for battery energy storage system safety management. It indicates that even after stopping the electrical abuse, the chemical abuse continues due to the reservoir of reactive lithium dendrites. The risk of progression to thermal failure after a shutdown is therefore directly related to the amount of these metastable materials present. The gas composition shift during full thermal runaway, with a significant increase in CH4, points to more severe cracking of electrolyte solvents at the highest temperatures.
Conclusions and Implications for Battery Energy Storage System Safety
This investigation into the overcharge-induced thermal failure of a large-format LFP cell yields several key conclusions for designing early warning systems in a battery energy storage system:
1. Voltage Warning Window: The effective warning period extends from the start of overcharge until the onset of the second, rapid voltage rise phase. Intervening by discharging or isolating the cell when the voltage reaches a critical threshold (e.g., 20 V for this cell, ~5.5 times nominal) can prevent thermal runaway. However, the exact threshold is cell-dependent and must be characterized for specific battery energy storage system modules.
2. Post-Intervention Risk: Halting overcharge does not immediately eliminate the risk. Exothermic chemical reactions continue, driven by residual lithium dendrites, causing a steady temperature increase. The cell remains in a precarious state, and any subsequent energy input can rapidly lead to failure. Safety protocols must account for this lingering hazard.
3. Optimal Warning Parameters: A multi-parameter warning strategy is most robust. Hydrogen (H2) is the premier early warning signal due to its earliest appearance and highest concentration. Carbon monoxide (CO) serves as a strong secondary gas warning. Among hydrocarbons, ethylene (C2H4) is a dominant and characteristic species. Voltage serves as a crucial backup and tertiary warning, especially in the late stages.
Based on our findings, we propose a tiered warning framework for a battery energy storage system:
Tier 1 (Early Stage): Primary reliance on cell voltage monitoring above the normal charge cutoff.
Tier 2 (Venting Stage): Activation of gas detection. H2 and CO detection trigger the primary alarm.
Tier 3 (Critical Stage): Detection of hydrocarbon gases (especially C2H4) and the onset of the second rapid voltage rise (e.g., >5.26 V climbing rapidly) trigger the highest-level alarm and immediate, forceful intervention such as module isolation and activation of suppression systems.
This work underscores that effective safety management for a battery energy storage system requires monitoring not just electrical parameters but also chemical signatures like gas evolution. Integrating real-time gas sensors with battery management systems can provide the critical minutes of advance warning needed to prevent a single cell failure from escalating into a catastrophic system-wide event.
