In the context of the rapid expansion of renewable energy, the solar system—encompassing photovoltaic panels, inverters, and balance-of-system components—plays a pivotal role in harnessing solar power. As a researcher focused on surface engineering for sustainable energy applications, I have extensively investigated advanced treatment techniques for aluminum alloy frames used in solar photovoltaic modules. These frames are critical structural elements that protect and support photovoltaic cells, directly influencing the longevity, efficiency, and reliability of the entire solar system. Traditional surface treatment methods, such as conventional anodic oxidation at elevated temperatures, often entail high energy consumption and yield inconsistent oxide film quality, posing challenges for the solar industry’s drive toward sustainability. This article delves into the low-temperature anodic oxidation process, a promising innovation for aluminum alloy frame treatment, analyzing its principles, characteristics, applications, and benefits for solar system integration. By leveraging this technology, we can enhance frame performance while reducing environmental impact, thereby supporting the global transition to clean energy.
The fundamental principle of anodic oxidation revolves around electrochemical reactions that form a protective oxide layer on aluminum surfaces. In this process, the aluminum alloy frame acts as the anode in an electrolytic cell, typically with a cathode made of lead or other inert materials. Upon applying a direct current, aluminum atoms at the anode lose electrons and dissolve as Al³⁺ ions into the electrolyte. Concurrently, oxygen-containing anions (e.g., sulfate or oxalate ions) migrate toward the anode, where they react with Al³⁺ and water molecules to form aluminum oxide (Al₂O₃). This oxide film exhibits a dual-layer structure: a thin, dense barrier layer adjacent to the metal substrate and a porous outer layer. The growth mechanism involves continuous oxidation and partial dissolution, governed by parameters like temperature, current density, and electrolyte composition. The reactions can be summarized as:
$$ \text{Anode: } 2Al + 3H_2O \rightarrow Al_2O_3 + 6H^+ + 6e^- $$
$$ \text{Cathode: } 6H^+ + 6e^- \rightarrow 3H_2 \uparrow $$
Low-temperature anodic oxidation, conducted typically between 5°C and 15°C, leverages reduced thermal energy to slow down ion diffusion and reaction kinetics. This deceleration fosters the formation of a more uniform, compact, and less porous oxide film compared to high-temperature processes. The enhanced film quality translates to superior hardness, corrosion resistance, and durability—attributes essential for solar system components exposed to harsh outdoor environments. Key advantages include energy savings due to lower heating requirements, improved film consistency, and reduced environmental footprint through minimized electrolyte waste. To optimize this process for solar system applications, factors such as electrolyte formulation, temperature control, current density, and oxidation time must be meticulously calibrated. For instance, sulfuric acid electrolytes at concentrations of 10–20% wt. are common, but organic acids like oxalic acid can yield thicker films at the cost of higher expense. The interplay of these parameters determines oxide film properties, which can be modeled using empirical relationships. For example, film thickness (δ) often correlates with current density (i) and time (t) via:
$$ \delta = k \cdot i \cdot t $$
where k is a material- and process-dependent constant. Below is a table summarizing the influence of key parameters on oxide film characteristics in low-temperature anodic oxidation:
| Parameter | Typical Range | Effect on Oxide Film | Implications for Solar System Frames |
|---|---|---|---|
| Temperature | 5–15°C | Lower temperature increases film density and hardness, but slows growth rate. | Enhances corrosion resistance for long-term outdoor exposure in solar systems. |
| Current Density | 1–2 A/dm² | Higher density accelerates growth but may cause roughness; lower density improves uniformity. | Ensures consistent protection across frame surfaces, vital for solar system reliability. |
| Electrolyte Concentration | 10–20% H₂SO₄ | Lower concentration yields denser films; higher concentration increases porosity. | Optimizes pore structure for subsequent sealing, boosting weatherability in solar systems. |
| Oxidation Time | 20–60 minutes | Longer time increases thickness, but excessive time may lead to film deterioration. | Balances film thickness (e.g., 10–25 μm) to meet mechanical and protective demands of solar systems. |
| Agitation | Moderate stirring | Improves heat and mass transfer, reducing localized overheating. | Maintains film homogeneity, critical for frames in large-scale solar system installations. |
Surface treatment requirements for aluminum alloy frames in solar systems are stringent due to the demanding operational conditions. These frames must exhibit excellent appearance quality, including smoothness, uniform coloration, and absence of defects like scratches or pits, to ensure aesthetic integration into diverse solar system architectures. More critically, they demand high corrosion resistance to withstand ultraviolet radiation, moisture, acid rain, and saline atmospheres—common in coastal or industrial solar system deployments. Additionally,耐磨性 is essential to endure mechanical abrasion from wind-blown sand, installation handling, and maintenance activities. Traditional treatments often fall short in these aspects, but low-temperature anodic oxidation addresses them effectively by producing a robust oxide layer. The corrosion resistance can be quantified using polarization resistance (R_p) derived from electrochemical impedance spectroscopy (EIS), where higher R_p values indicate better protection. For a solar system frame, the target R_p might exceed 10⁶ Ω·cm² after treatment, ensuring decades of service life.
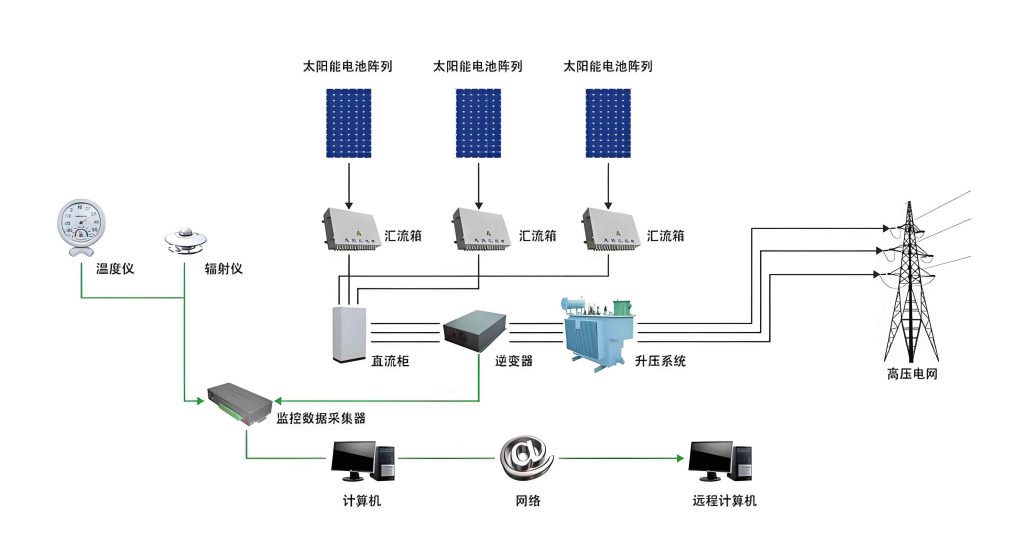
The application of low-temperature anodic oxidation to solar system aluminum alloy frames involves a structured workflow. Pre-treatment steps include alkaline degreasing to remove oils, followed by acid etching to eliminate native oxides, ensuring a clean substrate for oxidation. Subsequently, the frames are immersed in a chilled electrolyte bath and subjected to controlled DC power. Post-oxidation, sealing treatments—such as hot water sealing or nickel-free chemical sealing—fill the porous layer to enhance corrosion and abrasion resistance. Coloration using organic or inorganic dyes can be applied for aesthetic customization, aligning with solar system design requirements. Process optimization is crucial; for example, temperature must be maintained within ±1°C of the setpoint to avoid film defects. Current density profiles may be adjusted using pulse or constant voltage modes to improve film uniformity. The table below outlines a typical low-temperature anodic oxidation recipe for solar system frames:
| Process Stage | Conditions | Duration | Outcome |
|---|---|---|---|
| Degreasing | 5% NaOH at 50°C | 5–10 minutes | Removes contaminants for better adhesion. |
| Acid Etching | 10% HNO₃ at room temperature | 2–5 minutes | Activates surface for uniform oxidation. |
| Anodic Oxidation | 15% H₂SO₄ at 10°C, 1.5 A/dm² | 40 minutes | Forms 15 μm thick, dense oxide film. |
| Sealing | Nickel-free sealant at 80°C | 15 minutes | Reduces porosity, boosts corrosion resistance. |
| Drying | Hot air at 60°C | 10 minutes | Prevents water spots, ensures finish stability. |
To evaluate treatment efficacy, multiple assessment methods are employed. Appearance inspection via optical microscopy or spectrophotometry checks for color consistency and surface defects—critical for solar system components where visual uniformity matters. Corrosion resistance is tested through salt spray exposure per ASTM B117, where frames are subjected to 5% NaCl fog at 35°C; a high-performance solar system frame should withstand over 1000 hours without significant pitting or coating failure. Abrasion resistance can be measured using Taber abrasion tests, with weight loss after 1000 cycles ideally below 10 mg. Additionally, adhesion tests, such as cross-hatch adhesion per ISO 2409, ensure the oxide film remains intact under mechanical stress. These evaluations validate that low-temperature anodic oxidation meets the rigorous standards of solar system applications, often outperforming traditional methods. For instance, the hardness of the oxide film, measured by Vickers indentation, can reach 400–500 HV, compared to 300–350 HV for high-temperature oxides, directly benefiting solar system durability.
A case study from the solar industry illustrates the practical benefits. A photovoltaic manufacturer transitioning from conventional anodic oxidation to low-temperature anodic oxidation for its aluminum alloy frames observed marked improvements. Pre-treatment was refined with eco-friendly degreasers, and oxidation was conducted at 10°C with optimized current density profiles. Post-treatment included advanced sealing techniques. Results showed a 25% increase in oxide film density, a 30% enhancement in hardness, and a doubling of salt spray resistance to 1500 hours. Energy consumption dropped by 35%, and production cycle time reduced by 20%, lowering costs. The frames demonstrated superior performance in field tests across diverse solar system installations, from arid deserts to humid coastal regions, with no corrosion-related failures over two years. This case underscores how low-temperature anodic oxidation can elevate solar system reliability while supporting sustainability goals. The economic and environmental gains are quantifiable: for a solar system with 10,000 frames annually, energy savings exceed 50 MWh, and CO₂ emissions are cut by 15 metric tons, aligning with green manufacturing trends.
Further optimization of low-temperature anodic oxidation involves mathematical modeling and advanced control strategies. The growth kinetics of the oxide film can be described by a modified Deal-Grove model, accounting for low-temperature effects:
$$ \frac{d\delta}{dt} = \frac{A}{\delta} + B $$
where A and B are constants related to electric field and interfacial reactions. In solar system applications, maintaining consistent film properties across large frame batches requires real-time monitoring of electrolyte conductivity and pH, which can be automated using sensors and feedback loops. Additionally, the integration of renewable energy sources, such as solar-powered chilling units for temperature control, can further reduce the carbon footprint of the process. Research into novel electrolytes—like boric-sulfuric acid mixtures—promises even better film qualities at temperatures as low as 0°C, potentially revolutionizing frame treatment for extreme climate solar systems. The table below compares low-temperature anodic oxidation with traditional methods for solar system frames:
| Aspect | Traditional High-Temperature Anodic Oxidation | Low-Temperature Anodic Oxidation | Impact on Solar System Performance |
|---|---|---|---|
| Energy Consumption | High (heating to 20–25°C or above) | Low (cooling to 5–15°C) | Reduces operational costs and supports green solar system initiatives. |
| Oxide Film Density | Moderate, higher porosity | High, lower porosity | Enhances corrosion resistance, extending solar system lifespan. |
| Film Hardness (HV) | 300–350 | 400–500 | Improves abrasion resistance for frames in dusty solar system environments. |
| Process Consistency | Variable due to thermal fluctuations | Stable with precise temperature control | Ensures uniform quality across all frames in a solar system project. |
| Environmental Impact | Higher waste generation and emissions | Lower, with potential for electrolyte recycling | Aligns with sustainable solar system manufacturing practices. |
In conclusion, low-temperature anodic oxidation represents a transformative approach for treating aluminum alloy frames in solar systems. By operating at reduced temperatures, this process yields oxide films with exceptional density, hardness, and corrosion resistance—key attributes for withstanding the rigors of outdoor solar system deployments. Through meticulous parameter optimization, including electrolyte composition, current density, and sealing techniques, frames can achieve performance metrics that surpass traditional methods. The technology not only enhances product quality but also reduces energy consumption and environmental impact, contributing to the sustainability of the solar industry. As solar systems expand globally, adopting advanced surface treatments like low-temperature anodic oxidation will be crucial for ensuring long-term reliability and efficiency. Future research should focus on further lowering process temperatures, exploring eco-friendly electrolytes, and integrating smart manufacturing tools to maximize benefits for solar system applications. Ultimately, this innovation supports the vision of a cleaner energy future, where solar systems operate efficiently and durably for decades.
