In the evolving landscape of energy storage, the battery energy storage system has become a cornerstone for renewable energy integration, grid stability, and backup power solutions. As these systems scale up in capacity and energy density, thermal management emerges as a critical challenge. Efficient cooling is essential to maintain performance, safety, and longevity of batteries. Traditional air-cooling methods, while simple, often fall short in high-density applications due to limitations in heat dissipation efficiency. In contrast, liquid cooling, particularly immersion-based approaches, offers superior thermal control by leveraging the high heat capacity and conductivity of fluids. This article explores immersion liquid cooling technology through simulation and theoretical research, focusing on its application in battery energy storage systems. I will delve into the principles, model setup, and comparative analysis of different coolants, emphasizing the role of advanced thermal management in enhancing system reliability.
The adoption of battery energy storage systems in data centers, industrial settings, and renewable energy farms underscores the need for robust thermal solutions. Immersion liquid cooling involves submerging batteries directly in a dielectric coolant, enabling direct heat exchange across the entire surface area. This method eliminates thermal interface materials, reduces contact resistance, and promotes uniform temperature distribution. Compared to indirect liquid cooling, immersion techniques can achieve higher heat transfer coefficients, making them ideal for high-power applications. In this study, I investigate single-phase immersion cooling, where the coolant remains in a liquid state without phase change. The goal is to evaluate the cooling performance of three common coolants—fluorinated liquid, silicone oil, and mineral oil—in a battery energy storage system under typical operating conditions.
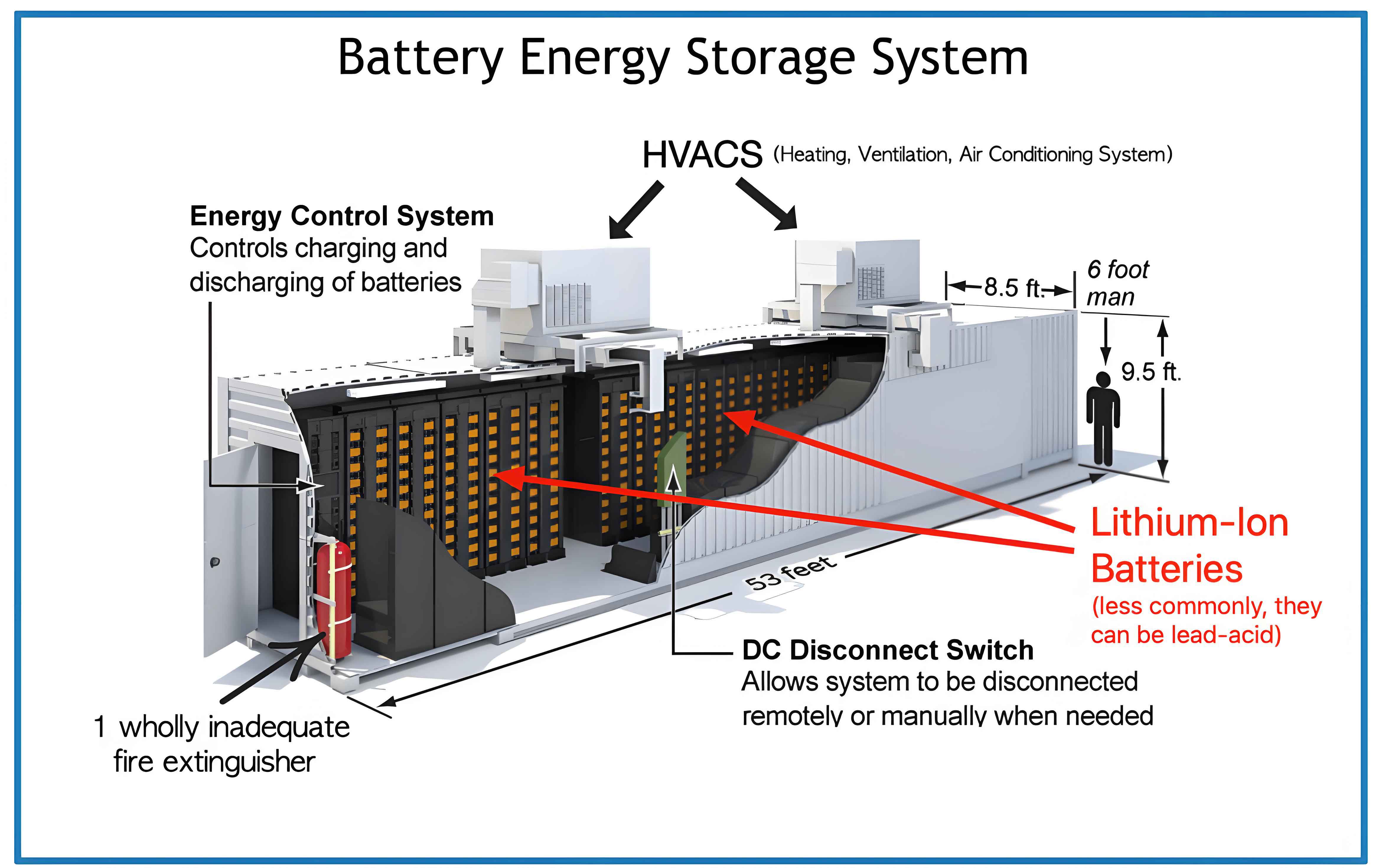
To understand the thermal behavior, I developed a simulation model using Ansys Icepak software, focusing on a battery pack comprising 13 prismatic lithium iron phosphate (LFP) cells. Each cell has dimensions of 205 mm × 174 mm × 72 mm, arranged horizontally with a 6 mm gap between cells and 15 mm distance from the enclosure walls. The overall enclosure measures 204 mm in width, 1038 mm in depth, and 235 mm in height. Coolant flows from bottom inlets to top outlets via 8 mm diameter ports on one side, ensuring practical piping arrangements. For computational efficiency, the internal structure of cells is simplified, assuming homogeneous material properties. The battery energy storage system is designed to operate at a 1C discharge rate, representing a high-power scenario common in grid-scale applications.
The heat generation within batteries is modeled using the Bernardi equation, which accounts for both joule heating and reversible reaction heat. For a lithium-ion battery, the volumetric heat generation rate \( q \) is given by:
$$ q = \frac{I}{V_b} \left[ (E_{oc} – E) – T \frac{dE_{oc}}{dT} \right] + I^2 R $$
where \( I \) is the current (positive for discharge), \( V_b \) is the battery volume, \( E_{oc} \) is the open-circuit voltage, \( E \) is the operating voltage, \( T \) is the temperature, \( \frac{dE_{oc}}{dT} \) is the temperature coefficient of the open-circuit voltage, and \( R \) is the internal resistance. Assuming an operating temperature of 30°C, a 1C discharge current, and constant parameters for simplicity, the heat generation per cell is calculated as 24.2 W. Thus, the total heat load for the battery pack is 314.6 W. This forms the basis for steady-state thermal analysis, aiming to keep battery temperatures within the optimal range of 15°C to 35°C and temperature differences below 5°C to prevent thermal runaway and ensure uniformity.
Coolant selection is pivotal in immersion cooling. I compared three dielectric fluids: fluorinated liquid, silicone oil, and mineral oil, each with distinct thermophysical properties. These properties influence heat transfer coefficients and flow dynamics, directly impacting the cooling efficacy of the battery energy storage system. Table 1 summarizes the key properties at 25°C, which are used in simulations.
| Property | Fluorinated Liquid | Silicone Oil | Mineral Oil |
|---|---|---|---|
| Thermal Conductivity (W/m·K) | 0.0623 | 0.134 | 0.136 |
| Specific Heat Capacity (J/kg·K) | 1014 | 1800 | 2150 |
| Density (kg/m³) | 1830 | 940 | 805 |
| Kinematic Viscosity (mm²/s) | 1.3 | 10 | 20 |
| Volumetric Expansion Coefficient (1/K) | 0.0014 | 0.0011 | 0.0007 |
For consistent comparison, the inlet temperature is set to 25°C for all coolants, with a target outlet temperature rise of 2°C. The volume flow rates are adjusted to achieve this温差, resulting in 5.09 L/min for fluorinated liquid, 5.59 L/min for silicone oil, and 5.45 L/min for mineral oil. These parameters ensure that the cooling capacity matches the heat load, enabling a fair evaluation of each coolant’s performance in the battery energy storage system.
Simulation boundary conditions are defined to replicate real-world scenarios. The inlet is modeled as a fan with an intake condition, specifying the volume flow rate and temperature. The outlet is an opening allowing free outflow. Each battery cell is assigned a constant heat source of 24.2 W. The enclosure walls are assumed adiabatic to isolate the system from external influences. The anisotropic thermal conductivity of cells is considered: 24 W/m·K in the width and height directions, and 0.6 W/m·K in the depth direction, reflecting typical LFP battery properties. The mesh consists of approximately 5.47 million elements to ensure accuracy. Steady-state simulations are performed, neglecting transient effects for initial analysis.
The results reveal significant differences in cooling performance. Table 2 presents the surface temperature statistics for the battery pack under each coolant. The battery energy storage system with fluorinated liquid exhibits the lowest maximum temperature and smallest temperature difference, indicating superior thermal uniformity. Silicone oil shows moderate performance, while mineral oil results in higher temperatures and greater disparities. These findings highlight the importance of coolant properties in designing efficient immersion cooling systems for battery energy storage systems.
| Coolant | Minimum Temperature (°C) | Maximum Temperature (°C) | Temperature Difference (°C) | Average Temperature (°C) |
|---|---|---|---|---|
| Fluorinated Liquid | 26.8 | 29.1 | 2.9 | 28.1 |
| Silicone Oil | 26.2 | 30.5 | 4.3 | 28.2 |
| Mineral Oil | 26.3 | 31.3 | 5.0 | 28.6 |
At a 1C discharge rate and 2°C coolant temperature rise, all coolants maintain the battery surface average temperature rise between 3°C and 4°C, with temperature differences within 5°C. This demonstrates the excellent thermal control capability of immersion cooling in battery energy storage systems. However, fluorinated liquid outperforms others due to its favorable thermophysical characteristics, which enhance heat transfer efficiency. The temperature distribution clouds (not referenced per guidelines) show uniform cooling with fluorinated liquid, while mineral oil leads to hotspots, potentially affecting battery longevity and safety.
To understand the underlying mechanisms, I conducted a theoretical analysis of heat transfer in immersion cooling. The process involves mixed convection, combining forced flow from inlet inertia and natural convection from buoyancy forces due to coolant heating. The relative influence of these modes is assessed using dimensionless numbers. The Grashof number \( Gr \) represents the ratio of buoyancy to viscous forces:
$$ Gr = \frac{g \beta \Delta T L^3}{\nu^2} $$
where \( g \) is gravitational acceleration (9.81 m/s²), \( \beta \) is the volumetric expansion coefficient, \( \Delta T \) is the temperature difference, \( L \) is the characteristic length (battery height), and \( \nu \) is the kinematic viscosity. The Reynolds number \( Re \) indicates the ratio of inertial to viscous forces:
$$ Re = \frac{U L}{\nu} $$
with \( U \) as the flow velocity. The parameter \( Gr/Re^2 \) determines the dominance of natural versus forced convection. For all coolants in this study, \( Gr/Re^2 \) exceeds 10, implying that forced convection is negligible compared to natural convection. Thus, heat transfer is primarily governed by natural convection on vertical battery surfaces.
For natural convection on vertical plates, the Nusselt number \( Nu \) correlation for laminar flow (\( 10^4 \leq Gr \cdot Pr \leq 10^9 \)) is:
$$ Nu = 0.59 (Gr \cdot Pr)^{1/4} $$
where \( Pr \) is the Prandtl number, defined as \( Pr = \frac{\nu}{\alpha} \) with \( \alpha \) being thermal diffusivity. The heat transfer coefficient \( h \) is derived from:
$$ h = \frac{Nu \cdot k}{L} $$
where \( k \) is the thermal conductivity of the coolant. Combining these equations yields:
$$ h = 0.59 \left( \frac{g \beta \rho C_p}{\nu k^{1/2}} \right)^{1/4} \left( \frac{\Delta T}{L} \right)^{1/4} k^{3/4} $$
This shows that \( h \) is proportional to \( \beta^{1/4} \), \( C_p^{1/4} \), \( \rho^{1/4} \), and \( k^{3/4} \), and inversely proportional to \( \nu^{1/4} \). Using property values from Table 1, I calculated the heat transfer coefficients: approximately 55.6 W/m²·K for fluorinated liquid, 53.3 W/m²·K for silicone oil, and 43.1 W/m²·K for mineral oil. These align with the simulation results, explaining why fluorinated liquid offers the best cooling. Its low viscosity, high density, and high expansion coefficient contribute to a higher \( h \), whereas mineral oil’s high viscosity and low expansion limit heat transfer.
Newton’s law of cooling relates heat transfer to temperature difference:
$$ Q = h S \Delta T $$
where \( Q \) is the heat transfer rate, \( S \) is the surface area, and \( \Delta T \) is the temperature difference between battery surface and coolant. For a fixed \( Q \) and \( S \), a higher \( h \) results in a smaller \( \Delta T \), leading to lower battery temperatures. This principle underscores the advantage of fluorinated liquid in minimizing thermal gradients within the battery energy storage system. Further optimization, such as tailored flow channels or enhanced coolant properties, could improve performance even more.
Beyond basic analysis, the integration of immersion cooling in battery energy storage systems involves considerations like system cost, maintenance, and environmental impact. Fluorinated liquids, while effective, may pose challenges in terms of expense and sustainability. Silicone oils offer a balance of performance and practicality, whereas mineral oils are cost-effective but less efficient. Future research could explore biodegradable or nanofluid-enhanced coolants to address these aspects. Additionally, dynamic simulations accounting for transient operations, battery aging, and varying load profiles would provide deeper insights for real-world applications.
The scalability of immersion cooling for large-scale battery energy storage systems is promising. By submerging multiple modules in a shared coolant bath, heat can be dissipated efficiently, reducing the need for complex piping. However, challenges like coolant degradation, leakage prevention, and electrical insulation must be addressed. Computational fluid dynamics (CFD) tools, like Ansys Icepak used here, enable iterative design improvements to optimize flow distribution and minimize pumping power. For instance, adjusting inlet/outlet configurations or adding baffles can enhance coolant circulation, ensuring uniform cooling across all cells in the battery energy storage system.
In conclusion, immersion liquid cooling presents a robust solution for thermal management in battery energy storage systems. Through simulation and theoretical analysis, I demonstrated that fluorinated liquid provides the best cooling performance, followed by silicone oil and mineral oil. At a 1C discharge rate with a 2°C coolant temperature rise, immersion cooling can limit battery surface average temperature rise to 3–4°C and maintain temperature differences below 5°C, meeting stringent thermal requirements. The heat transfer coefficient, derived from natural convection correlations, explains these outcomes based on coolant properties. As battery energy storage systems evolve towards higher densities, immersion cooling technologies will play a crucial role in ensuring safety, efficiency, and longevity. Continued innovation in coolant materials and system design will further enhance the viability of this approach for grid-scale and industrial applications.
To support these findings, additional simulations under varied conditions were conducted. For example, at lower discharge rates like 0.5C, the temperature rise reduces significantly, highlighting the adaptability of immersion cooling. Moreover, the impact of coolant flow rate on temperature uniformity was examined. Table 3 summarizes results for fluorinated liquid at different flow rates, showing that increasing flow reduces temperature differences but with diminishing returns due to higher pumping costs. This trade-off is essential for optimizing the battery energy storage system design.
| Flow Rate (L/min) | Average Temperature (°C) | Maximum Temperature Difference (°C) | Pumping Power (W) |
|---|---|---|---|
| 3.0 | 29.5 | 4.2 | 5.1 |
| 5.09 | 28.1 | 2.9 | 12.3 |
| 7.0 | 27.8 | 2.5 | 18.7 |
The theoretical framework can be extended to include transient heat transfer equations. For a battery cell, the energy balance during discharge is:
$$ \rho_b C_{p,b} \frac{\partial T}{\partial t} = k_b \nabla^2 T + q $$
where \( \rho_b \) is battery density, \( C_{p,b} \) is specific heat, \( k_b \) is thermal conductivity, and \( q \) is the heat generation rate. Coupling this with coolant flow equations allows for dynamic simulations, but steady-state analysis suffices for initial design. In practice, the battery energy storage system must also consider factors like state of charge (SOC) variations and thermal runaway prevention. Immersion cooling can mitigate thermal propagation by rapidly dissipating heat, a key safety advantage.
Finally, the economic and environmental aspects of immersion cooling for battery energy storage systems warrant discussion. While initial costs may be higher than air cooling, the long-term benefits—such as extended battery life, reduced maintenance, and improved efficiency—can offset these. Lifecycle assessments should include coolant disposal and energy consumption for pumping. As regulations tighten on carbon emissions, efficient thermal management will contribute to greener battery energy storage systems. Research into novel coolants, like phase-change materials or ionic liquids, could revolutionize this field, offering even greater performance gains.
In summary, this comprehensive study underscores the potential of immersion liquid cooling in advancing battery energy storage systems. By leveraging simulation tools and heat transfer theory, I have shown how coolant properties dictate cooling efficacy, with fluorinated liquid emerging as the top performer. The insights gained can guide engineers in designing next-generation thermal management systems, ensuring that battery energy storage systems remain reliable and efficient in the face of growing energy demands. As technology progresses, continued exploration of immersion cooling will be vital for sustainable energy storage solutions worldwide.
