The global transition towards renewable energy has been significantly accelerated by the widespread deployment of Battery Energy Storage Systems (BESS). These systems are pivotal for grid stability, load shifting, and integrating intermittent sources like solar and wind. However, a critical environmental challenge emerges at the end of their service life. The solid waste generated from decommissioned batteries, containing toxic organic solvents and corrosive lithium salts, presents a substantial pollution risk if not managed with utmost rigor. This article, from my professional perspective as an environmental engineer specializing in industrial waste management, delves into a systematic framework for mitigating these risks, moving beyond traditional, often inadequate, containment methods.
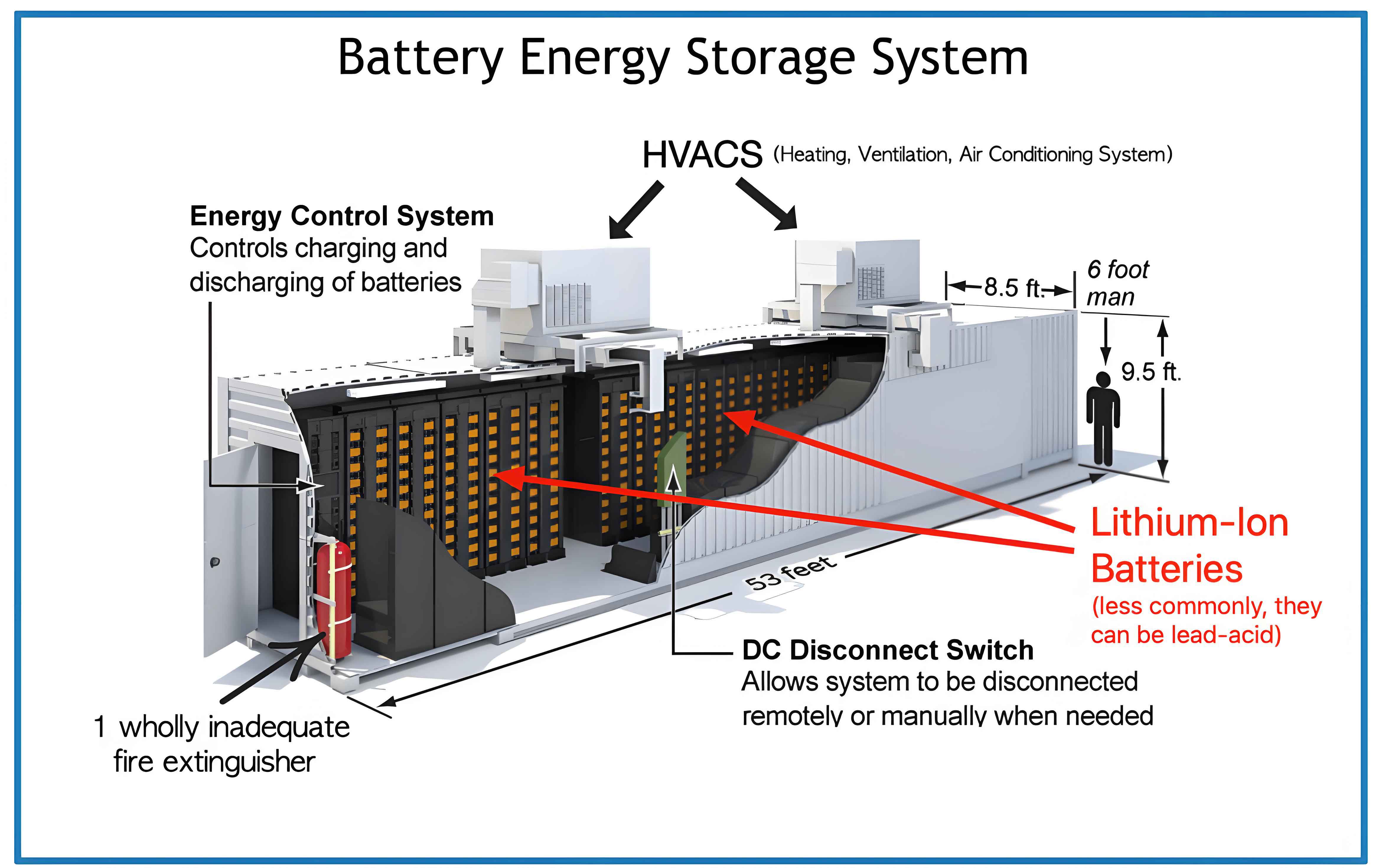
The core of the problem lies in the chemistry of the dominant battery chemistries within a typical BESS, primarily Lithium Iron Phosphate (LFP) and Nickel Manganese Cobalt (NMC) types. Upon retirement and during storage prior to recycling, physical damage or gradual degradation can lead to the leakage of electrolyte. This electrolyte is typically a complex mixture of organic carbonates (e.g., ethylene carbonate, dimethyl carbonate) and a conductive lithium salt, most commonly lithium hexafluorophosphate (LiPF6). The environmental hazards are twofold: the organic solvents can contaminate soil and groundwater, while LiPF6 reacts readily with moisture to generate highly corrosive and toxic hydrofluoric acid (HF). Furthermore, damaged cells pose a thermal runaway risk, potentially releasing a cocktail of hazardous gases. Therefore, the interim storage and handling of solid waste from a Battery Energy Storage System demand a scientifically robust, multi-barrier approach to prevent the migration of pollutants into soil, water, and air.
My analysis is grounded in the observed shortcomings of conventional storage practices at electrochemical energy storage facilities. The traditional model often relies on single-layer clay liners or ordinary concrete flooring for containment, supplemented by simple bunds and natural ventilation. While these provide a basic level of isolation, long-term performance is questionable. Monitoring data from such facilities frequently reveals insidious, low-level contamination, indicating that pollutants are gradually breaching these primary defenses. The migration of contaminants can be modeled using a simplified advection-dispersion equation adapted for porous media like soil under a waste pile:
$$ \frac{\partial C}{\partial t} = D \frac{\partial^2 C}{\partial x^2} – v \frac{\partial C}{\partial x} – \lambda C $$
Where \( C \) is the contaminant concentration (e.g., fluoride ions, organic compounds), \( D \) is the hydrodynamic dispersion coefficient, \( v \) is the average linear groundwater velocity, and \( \lambda \) is a first-order decay constant. A high-performance lining system aims to reduce \( v \) to an effectively negligible value, thereby stifling the advective term \( -v \frac{\partial C}{\partial x} \). Similarly, for airborne risks, the accumulation of gases like HF in a storage shed depends on the generation rate and the ventilation rate. Natural ventilation, being passive and variable, often fails to maintain a safe dilution. A forced ventilation system ensures a controlled, high air exchange rate, which can be described by a mass balance for a well-mixed air volume:
$$ V \frac{dC_{air}}{dt} = G – Q C_{air} $$
At steady state (\( dC_{air}/dt = 0 \)), the indoor concentration \( C_{air} \) is given by \( C_{air} = G / Q \), where \( V \) is the shed volume, \( G \) is the gas generation rate, and \( Q \) is the volumetric ventilation flow rate. Increasing \( Q \) through mechanical means directly reduces \( C_{air} \). These principles form the theoretical foundation for the multi-level control framework I advocate for managing solid waste from any large-scale Battery Energy Storage System.
Characteristics and Hazards of Solid Waste from Battery Energy Storage Systems
A thorough risk assessment must begin with a precise understanding of the waste stream. The solid waste from a decommissioned Battery Energy Storage System is not homogeneous. It primarily consists of the battery modules and packs, which house the cells, wiring, and battery management systems. The principal environmental vectors are the electrochemical components within the cells. The table below summarizes the key hazardous constituents and their associated risks for the two prevalent chemistries.
| Battery Chemistry | Key Hazardous Components in Solid Waste | Primary Environmental Risks | Notes on Stability |
|---|---|---|---|
| Lithium Iron Phosphate (LFP) | Electrolyte: LiPF6 in organic carbonates. | Soil/water contamination from solvents; HF generation upon moisture ingress. | Thermally more stable, lower fire risk, but electrolyte hazards remain identical. |
| Nickel Manganese Cobalt (NMC) | Electrolyte: LiPF6 in organic carbonates. Cathode contains Ni, Co, Mn. | All risks from LFP, plus potential leaching of heavy metals (Ni, Co). Higher thermal runaway risk. | Higher energy density correlates with higher reactivity and more severe fire/gas emission risk. |
The risk is dynamic. A mechanically compromised cell allows electrolyte to seep out. LiPF6 hydrolysis is a quintessential hazard, proceeding as:
$$ \text{LiPF}_{6(s)} + \text{H}_2\text{O} \rightarrow \text{LiF}_{(s)} + \text{POF}_{3(g)} + 2\text{HF}_{(g)} $$
The generated phosphorus oxyfluoride (POF3) further hydrolyzes to produce additional HF. Therefore, managing moisture is as critical as managing the physical leakage. Furthermore, in a fire scenario, the decomposition pathways become more complex, yielding a wide range of toxic organofluorine compounds and particulates. The solid waste storage facility for a Battery Energy Storage System must therefore be designed to address three primary exposure pathways: Leachate to soil and groundwater, emissions to the atmosphere, and acute spillage to the surrounding area.
A Multi-Level Risk Prevention and Control Framework
Addressing the complex, multi-pathway risks requires a defense-in-depth strategy. The proposed framework is structured into three hierarchical, interconnected levels: Primary Containment (Source Isolation), Secondary Control (Environmental Mitigation), and Tertiary Defense (Emergency Response). This structure ensures that if one barrier is challenged or breached, subsequent barriers are in place to prevent environmental release.
The efficacy \( E \) of this multi-barrier system can be conceptually framed as a series system where the overall probability of failure \( P_f \) is the product of the failure probabilities of each independent barrier:
$$ P_f = p_1 \times p_2 \times p_3 $$
$$ E = 1 – P_f $$
Here, \( p_1, p_2, p_3 \) represent the probability of failure for the primary, secondary, and tertiary barriers, respectively. The goal of engineering design is to drive each \( p_i \) as close to zero as possible, making \( P_f \) exceedingly small. The following sections detail the specific measures constituting each barrier, tailored for the solid waste from a Battery Energy Storage System.
Level 1: Primary Containment – Impermeable Storage Measures
The first and most crucial line of defense is to physically encapsulate the waste and prevent any liquid or solid contaminants from entering the natural environment. This involves engineered structures with exceptionally low permeability. The design moves far beyond simple clay liners.
1. Composite Liner System: The floor of the designated storage building or pad must be a composite liner. The optimal configuration consists of:
- A primary liner of high-density polyethylene (HDPE) geomembrane, minimum 2.0 mm thick, with all seams thermally welded and tested.
- A secondary liner of compacted clay or a synthetic clay liner (GCL), providing additional adsorption capacity and redundancy.
- A protective layer of geotextile and a drainage layer (leachate collection system) above the liners.
- A top layer of chemical-resistant, reinforced concrete with a permeability coefficient \( k \leq 1 \times 10^{-12} \) m/s, sloped at a minimum gradient of 2% towards collection points.
The performance of this composite barrier is quantified by its hydraulic conductivity. The combined effect can be evaluated using a series flow model for layered systems. The effective vertical hydraulic conductivity \( K_{v(eff)} \) for \( n \) layers is given by:
$$ K_{v(eff)} = \frac{d}{\sum_{i=1}^{n} (d_i / K_{v_i})} $$
where \( d \) is the total thickness and \( d_i \) and \( K_{v_i} \) are the thickness and vertical hydraulic conductivity of the i-th layer. By selecting materials with extremely low \( K_{v_i} \) (e.g., HDPE \( K_v \) ~ \( 1 \times 10^{-14} \) m/s, compacted clay \( K_v \) ~ \( 1 \times 10^{-9} \) m/s), the \( K_{v(eff)} \) of the entire composite system is rendered minimal, drastically reducing the Darcy flux \( q = K \cdot i \) (where \( i \) is the hydraulic gradient).
2. Integral Bunding and Segregation: The entire concrete pad must be surrounded by an integral bund wall. Its capacity should be at least 110% of the volume of the largest storage container or 25% of the total waste volume stored on the pad, whichever is greater. Internally, the pad should be subdivided using low curbs to segregate different waste streams (e.g., intact modules, visibly damaged cells, defective units from the Battery Energy Storage System). Each sub-cell should have independent drainage towards the central collection system. All stored waste must be placed on secondary containment pallets or within spill decks.
3. Leachate Collection and Detection: The slope of the floor directs any seepage to perimeter channels or a central sump connected to a dedicated leachate collection tank. This system must include a leak detection layer between the primary and secondary liners, capable of triggering an alarm upon sensing moisture. The collected leachate, high in fluorides and organics, must be treated as hazardous liquid waste and processed accordingly—typically through chemical precipitation (for fluorides) and advanced oxidation or carbon adsorption (for organics) before any discharge.
| Component | Design Specification | Performance Target | Function |
|---|---|---|---|
| HDPE Liner | >= 2.0 mm thickness, certified material | Permeability <= 1×10-14 m/s | Primary hydraulic barrier |
| Compacted Clay / GCL | Min. 0.6 m compacted clay or equivalent GCL | Permeability <= 1×10-9 m/s | Secondary barrier & adsorption |
| Chemical-Resistant Concrete | Reinforced, with additives for acid resistance | Permeability <= 1×10-12 m/s, Slope >= 2% | Wear surface & directed drainage |
| Bund Wall | Height >= 0.3 m, integral with floor | Containment volume >= 110% largest container | Contain large spills and firewater |
| Leachate Sump | Fiberglass or coated steel, with level sensor | Automatic pump transfer at high level | Collect and remove contaminated liquids |
Level 2: Secondary Control – Forced Ventilation and Air Quality Management
While primary containment addresses liquid and solid migration, it does not mitigate airborne risks from volatile organic compounds (VOCs) and gases like HF. A storage building for Battery Energy Storage System waste cannot rely on natural ventilation. A mechanically assisted, monitored, and treated ventilation system is non-negotiable.
1. Dilution Ventilation Design: The ventilation system must maintain the building under negative pressure relative to the outside to prevent the exfiltration of contaminated air. Air change rates (ACH) should be high, typically 6-12 ACH for general storage areas and potentially higher near designated inspection or handling zones. The required exhaust flow rate \( Q \) can be calculated based on the target indoor concentration \( C_{target} \) (set below occupational exposure limits like 0.5 ppm for HF) and an estimated contaminant generation rate \( G \):
$$ Q = \frac{G}{C_{target} – C_{background}} $$
Since \( G \) is highly variable and often unknown, a conservative approach based on ACH is used: \( Q = ACH \times V_{building} \). Intake vents should be positioned low on one side, and exhaust ducts high on the opposite side to promote cross-flow and prevent short-circuiting.
2. Exhaust Air Treatment: Simply exhausting contaminated air is unacceptable. The exhaust stream must be treated before release to the atmosphere. The most effective and proven technology for acid gases like HF is a packed-bed scrubber. The exhaust air is passed through a tower packed with plastic media while an alkaline solution (typically sodium hydroxide, NaOH) is sprayed counter-currently. The acid gas absorption is a mass transfer process. The removal efficiency \( \eta \) for a scrubber can be estimated using the following relationship, dependent on the number of transfer units (NTU):
$$ \eta = 1 – e^{-NTU} $$
$$ NTU = \frac{K_{og} \cdot a \cdot V}{Q} $$
where \( K_{og} \) is the overall mass transfer coefficient, \( a \) is the specific surface area of the packing, \( V \) is the scrubber volume, and \( Q \) is the gas flow rate. Properly designed, a scrubber can achieve >99% removal efficiency for HF. For VOCs, an additional stage like activated carbon filtration may be required downstream.
3. Continuous Atmospheric Monitoring: Real-time gas detection sensors are mandatory. These should be strategically placed at potential low points and accumulation zones (as HF is denser than air). Monitored parameters must include:
- Hydrogen Fluoride (HF): Primary acute hazard.
- Volatile Organic Compounds (VOCs): As a surrogate for solvent vapors.
- Carbon Monoxide (CO): Indicator of smoldering or thermal events.
- Combustible Gas (LEL): For fire risk warning.
Alarms must be tiered: a warning level (e.g., 0.5 ppm HF) triggers increased ventilation rates and an operator alert; an action level (e.g., 2 ppm HF) triggers evacuation alarms, shutdown of non-essential equipment, and activation of secondary scrubber pumps. Data from all sensors should be logged and trended for predictive maintenance and incident analysis.
Level 3: Tertiary Defense – Emergency Response and Collection Systems
This level addresses unforeseen failure modes or accident scenarios, such as a catastrophic rupture of a large module, a forklift incident, or a fire. Its purpose is to contain, control, and collect the released materials to prevent them from leaving the site boundary.
1. Fire Suppression and Containment: A dedicated fire protection system is essential. Due to the lithium metal and Class B fire risk, water-based suppression is ineffective and dangerous for large lithium fires. The design should incorporate:
- Early Detection: Aspirating smoke detectors (VESDA) and thermal cameras for rapid heat detection.
- Specialized Suppression: Automatic or manually applied Aerosol generators, or clean agent systems (e.g., NOVEC 1230) for electrical/control rooms. For storage areas, a deluge system may be considered to cool adjacent modules and prevent fire spread, but only in conjunction with a strategy for managing the resulting contaminated firewater.
- Containment of Runoff: The bunded area of the primary containment is the first line for holding firefighting water. The site must also have a plan and capacity (e.g., retention basins) to contain all firewater runoff, which will be heavily contaminated with fluorides, metals, and combustion byproducts.
2. Spill Kits and Neutralization: Strategically placed spill response stations must contain:
- Acid-neutralizing agents (e.g., calcium carbonate or sodium bicarbonate-based spill sorbents).
- Universal sorbent pads and booms for organic solvents.
- Personal protective equipment (PPE) including full-face respirators with acid gas cartridges, chemical suits, and gloves.
- Dedicated, labeled containers for collected spill waste.
The neutralization reaction for hydrofluoric acid with calcium carbonate, for instance, is:
$$ 2\text{HF} + \text{CaCO}_3 \rightarrow \text{CaF}_{2(s)} + \text{CO}_{2(g)} + \text{H}_2\text{O} $$
The precipitated calcium fluoride is relatively inert and can be disposed of as solid hazardous waste.
3. Emergency Procedures and Training: Engineering controls are futile without proper human response. Detailed Standard Operating Procedures (SOPs) must cover spill response, fire emergency, evacuation, and first-aid (specifically for HF exposure, requiring calcium gluconate gel). All personnel must undergo regular, hands-on training. Drills should simulate scenarios like a leaking Battery Energy Storage System module or a smoke alarm.
Empirical Validation and Performance Analysis
To quantify the effectiveness of the proposed multi-level framework compared to traditional methods, a controlled field trial can be conceptualized. Imagine a storage facility divided into two isolated sections: a Control Zone employing traditional single-layer concrete and natural ventilation, and an Intervention Zone implementing the full multi-level system (composite liner, forced ventilation with scrubbing, full monitoring). Both zones store identical volumes and types of decommissioned Battery Energy Storage System packs, including some intentionally damaged cells to simulate real-world conditions.
Key environmental indicators are monitored over a 180-day period:
- Soil/Sediment: Core samples taken beneath each zone’s liner (or at a shallow depth below traditional concrete) analyzed for Fluoride (F–), Total Organic Carbon (TOC), and heavy metals (Ni, Co).
- Leachate/Analyte: Any liquid collected in sumps or drainage channels analyzed for pH, F–, COD, and metals.
- Air Quality: Continuous monitoring of HF and VOC levels within the storage volume and at the exhaust stack (pre- and post-scrubber in the Intervention Zone).
The results would starkly illustrate the performance gap. The Control Zone would likely show a gradual increase in sub-surface contaminant concentrations and periodic spikes in indoor air contaminants during warm, low-wind days. The Intervention Zone would maintain background-level contaminant readings in the sub-liner environment and consistently low, compliant emissions.
| Performance Metric | Regulatory Limit | Control Zone (Traditional) | Intervention Zone (Multi-Level Framework) | Analysis |
|---|---|---|---|---|
| Soil Fluoride (mg/kg) Below containment |
400 (Typical industrial threshold) | 285 | < 5 (Detection Limit) | Composite liner effectively prevents vertical migration. |
| Leachate/Collection Sump Fluoride (mg/L) | 15 (Wastewater discharge limit) | 45.2 (Requires treatment) | 8.1 (Collected, requires less intensive treatment) | Collection system captures leakage; initial concentration indicates source containment is not perfect but controlled. |
| Indoor Air HF (ppm) – Peak | 0.5 (8-hr TWA) | 1.8 (Alarm condition) | 0.3 | Forced ventilation maintains dilution below action levels. |
| Exhaust Stack HF (ppm) – Average | 0.1 (Ambient air quality guideline) | 0.9 (Direct violation) | 0.02 | Scrubber achieves >97% removal efficiency, ensuring compliant emissions. |
| Chemical Oxygen Demand (COD) in Runoff Simulation (mg/L) | 50 | 122 | 22 | Bunding and spill management prevent organic solvent spread. |
The data supports the conclusion that a systematic, multi-barrier approach transforms the environmental risk profile of storing solid waste from a Battery Energy Storage System. The incremental capital and operational costs of such a system are justified not only by regulatory compliance but also by the avoidance of long-term liability from soil and groundwater remediation, which can be orders of magnitude more expensive.
Future Perspectives and Integration with Circular Economy
The ultimate goal for end-of-life Battery Energy Storage System components is not merely safe disposal, but high-value recycling. The risk防控 measures discussed herein are for the interim storage phase. The design of storage facilities should be forward-thinking to facilitate, not hinder, the recycling process. This includes:
1. Design for Disassembly (DfD): Future Battery Energy Storage System designs should consider ease of disassembly to reduce the hazard during the dismantling phase. This would minimize the number of damaged cells entering storage.
2. Co-location with Pre-processing: Ideally, storage facilities should be integrated with or located adjacent to pre-processing plants where batteries can be discharged, electrolyte can be safely extracted under controlled conditions, and modules can be mechanically broken down. This reduces the volume and hazard of the material needing long-term storage.
3. Advanced Monitoring and AI: The integration of IoT sensors, combined with artificial intelligence for predictive analytics, can elevate risk management. AI algorithms could analyze temperature trends from thermal cameras to predict thermal runaway events days in advance, or correlate VOC sensor data with barometric pressure to optimize ventilation cycles.
4. Lifecycle Risk Quantification: A holistic model can be developed to quantify the total environmental risk \( R_{total} \) across the lifecycle of a Battery Energy Storage System, including the waste phase. This could be expressed as a function of the effectiveness of each barrier:
$$ R_{total} = f(E_{primary}, E_{secondary}, E_{tertiary}, M, t) $$
where \( M \) is the mass and hazard potential of the waste, \( t \) is the storage time, and the \( E \) terms represent the efficacy of the multi-level barriers as previously defined. Optimizing this function through engineering and logistics minimizes the system’s overall environmental footprint.
Conclusion
The sustainable growth of the battery energy storage system industry is inextricably linked to solving its end-of-life environmental challenges. The solid waste generated poses real and significant risks to soil, water, and air quality. Relying on traditional, passive containment methods is insufficient and can lead to chronic low-level pollution and acute incident hazards. The multi-level prevention and control framework proposed—integrating engineered primary containment with composite liners, active secondary control through forced and treated ventilation, and robust tertiary emergency response systems—provides a comprehensive, defensible solution. This systematic approach, grounded in environmental engineering principles and supported by empirical performance data, ensures that the benefits of Battery Energy Storage System technology are not offset by its waste legacy. As the volume of decommissioned systems grows exponentially in the coming decade, the implementation of such rigorous standards will be critical for the truly sustainable development of the energy storage sector.
